4. EXPERIMENTAL PROCEDURE
4.6. Practice 4: Elimination of carbon dioxide in a column filler
Introduction
The aim of this practice is to clean the water of carbon dioxide that accumulates when were performed experiments with the absorption of this gas. Using analytical measurements we can determine the concentration of carbon dioxide in ppm and values obtained are compared with the solubility of CO2 in water, to know how much CO2 can absorb more water.
Materials and reagents
- A flask of 100 ml.
- Burette graduated.
- Flask of 250 ml.
- Apparatus agitator.
- Magnet.
- Tweezers.
- Shows water problem.
- Kit/test to measure the carbonate hardness (KH).
- 5 ml glass tube.
- Solution of sodium hydroxide (NaOH) 0.02 N.
- Phenolphthalein indicator solution.
Procedure
The procedure to perform the cleaning water is as follows:
- Supply water supply to the second pump (water jet pump).
- Close the valve pressure pump 2 (7).
- Close the inlet valve to the tank of depression (3).
- Adjust the water flow to pump water jet through the valve adjustment (8) (taking into account the manometer (2) the deposit of depression).
- Open a little valve (3) to the depression valve and carry water from the reservoir tank to depression tank (4), until the bottom of the tank is covered with approximately 5 cm of water .
- Connect the dual pushbutton pump 2 (1).
Figure 4.31: Distribution box.
Figure 4.32: Supply water.
Figure 4.33: Bottom of the device (1).
Figure 4.34: Bottom of the device (2).
1- Double pushbutton pump 2.
2- Pressure gauge.
3- Valve into the tank of depression.
4- Depression tank.
5- Depression pump 2.
6- Purge valve of the depression tank.
7- Pressure valves.
8- Valve regulated.
9- Thermometer.
10 - Pump 1 (pumping water).
11- Sample point (POINT 1)
The steps are:
- Open the valve pressure pump 2 (7), until the water level in the depression tank is constant (4).
- Control the depression tank (4), through the manometer (2). Whether to make changes can be made from water flow pump 2 or the inlet valve to the tank (3).
- In the event that the deposit of depression is very full, you must open the purge valve of the reservoir pressure depression (6).
- To determine if the water is clean enough, we analyzed with analytical methods.
The deposit of depression can work in parallel with the absorption of gases.
Then finish off the system and cleaning:
- Close to the water supply pump water jet.
- Close the inlet valve to the tank of depression (3).
- Disconnect the pump twin pushbutton 2 (1).
- Through the vent beneath the deposit of depression (four), evacuate the remaining water tank depression.
- For only the cleaning process is running, you must:
* Disconnect the general switch.
- In the case that all equipment is in operation, must:
* Disconnect the supply of CO2.
* Disconnect the water pump (pump 1), through the double push.
* Logout button twice the compressor.
* Disconnect the main switch.Methods for determination of carbon dioxide in water
Determination of carbonates with a rating of NaOH.
Consist in to react carbon dioxide content in a sample of water with sodium hydroxide (NaOH), thanks to the presence of an indicator (phenolphthalein). Causes the water sample increase its pH to a value of 10, so his pH varies an acid pH to a basic pH.
Below is the description of the material and the procedure to be followed for the assessment of the water sample with sodium hydroxide
Material
- A flask of 100 ml.
- Burette graduated.
- Flask of 250 ml.
- Apparatus agitator.
- Magnet.
- TweezersReagents
- Solution of sodium hydroxide (NaOH) 0.02 N.
- Phenolphthalein indicator solution.Procedure
- Take a water sample from the reservoir through the faucet purge. Placing the rubber tap to the bottom of the sample of 100 ml, so that no bubbles form.
- Transfer of the flask water 250 ml and add 10 drops of phenolphthalein indicator. If the sample bends pink indicates no presence of carbon dioxide free.
- In the case of the sample remains colourless, you must have a rating of 0.02 N sodium hydroxide Add drip solution valued by the burette until the Air shows a pink colour.
- We analyze the water before cleaning it and after cleaning it.
- All tests were performed in triplicate.
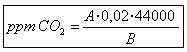
Where :
A: ml NaOH are spent in making the assessment.
B is the initial sample volume (100 ml).Determine the amount of CO2 that is water with Kit.
Here is the materials and procedure to follow to perform this test. Used to determine the amount of CO2 in water.
Material
- 5 ml glass tube.
- pHmetry.Reagents
- Show water problem.
- Kit / test to measure the carbonate hardness (KH).Procedure
- Collect a water sample from the reserve water tank using the tap purge. Placing the rubber tap the bottom of the beaker, so that no bubbles form.
- Measure the pH of the sample
- Take 5 ml sample and add a drop test kit (KH).
- In case turns blue, indicating that the sample has no carbonate hardness. Conversely, if is yellow, then drops should be added to get the colour blue. It is important to shake the tube every time you add a drop. The number of drops added KH coincide with the degree of the sample.
From the following formula can calculate the amount of carbon dioxide is present water
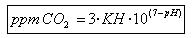
Where:
KH: number of drops added test.
pH: the number that provides pHmetry
Then we can see the video of the performance practice:
Questions
1) What is a reaction with carbon dioxide to mix with the sodium hydroxide?
2) What amount of sodium hydroxide is needed for preparing a solution of 0.02 N NaOH?
3) What is carbon dioxide concentration in the water before cleaning it?
4) What concentration of carbon dioxide there is in the water after cleaning it?
5) What is the solubility of CO2 in water?
6) Compare the value of the concentration of carbon dioxide before cleaning it with the value of the solubility of CO2. It can absorb more CO2 this water?

